
Some might expect such an ordered structure to be less stable because the entropy of the system would be low. Lattice Energy is used to explain the stability of ionic solids. Its values are usually expressed with the units kJ/mol. As implied in the definition, this process will always be exothermic, and thus the value for lattice energy will be negative. The other definition says that lattice energy is the reverse process, meaning it is the energy released when gaseous ions bind to form an ionic solid. This definition causes the value for the lattice energy to always be positive, since this will always be an endothermic reaction. In one definition, the lattice energy is the energy required to break apart an ionic solid and convert its component atoms into gaseous ions. Lattice Energy is a type of potential energy that may be defined in two ways. However, the basic difference between them is how they are calculated and what factors they take into account.\( \newcommand\) Lattice energy and Lattice Enthalpy both are measures to explain the stability of an ionic compound. Difference Between Lattice Energy and Lattice Enthalpy The Born-Haber cycle allows for the prediction of lattice energies for ionic compounds that have not yet been synthesized, as well as the determination of lattice energies for known compounds. Lattice Energy = Enthalpy of Formation + Electronic Affinity + Ionization Energy – Enthalpy of Sublimation of Positive Ion – Enthalpy of Vaporization of Negative ion Lattice energy can then be calculated using the following equation: This is the energy required to vaporize the negative ion. Calculating the enthalpy of vaporization of the negative ion.This is the energy required to vaporize the positive ion. Determining the enthalpy of sublimation of the positive ion.This is the energy required to remove an electron from a neutral atom to form a positive ion. Calculating the ionization energy of the positive ion.This is the energy required to add an electron to a neutral atom to form a negative ion. Calculating the electron affinity of the negative ion.This can be done through calorimetry or other methods. Determining the enthalpy of formation of the compound from its elements.Born-Haber cycle involves several steps, including: The lattice energy of an ionic compound is the energy required to separate the compound into its gaseous ions. The Born-Haber cycle is a method used to calculate the lattice energy of an ionic compound from experimentally determined or theoretically predicted quantities. n is Born Exponent which is different for Different MoleculesĬalculation of Lattice Energy using the Born-Haber Cycle.e is basic electronic charge = 1.6×10 -19 C.Z + and Z – is charge for cation and anion.As in this case, the lattice energy is the energy given to the ionic compound so this process is an endothermic reaction. The energy required to convert one mole of an ionic solid into its gaseous ionic constituents is called the lattice energy of the ionic compound. Ionic compounds have high melting and high boiling points due to the strong attractive forces between the ions. It is also related to the melting and boiling points of ionic compounds. The value of lattice energy can be used to predict the stability of an ionic compound and to compare the relative stabilities of different ionic compounds. We use various methods, such as the Born-Haber cycle or the Coulombic equation to find the lattice energy of any compound. The lattice energy of any compound is the energy given to a solid ionic compound to break into its gaseous state, we can also say that it is the amount of energy released when gaseous ions combine to form a solid lattice. Software Engineering Interview Questions.Top 10 System Design Interview Questions and Answers.Top 20 Puzzles Commonly Asked During SDE Interviews.Commonly Asked Data Structure Interview Questions.Top 10 algorithms in Interview Questions.Top 20 Dynamic Programming Interview Questions. 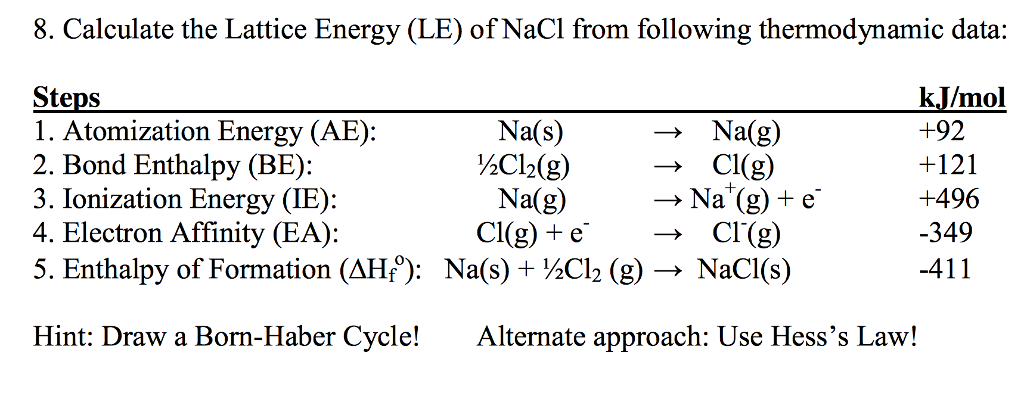
Top 20 Hashing Technique based Interview Questions.Top 50 Dynamic Programming (DP) Problems.Top 20 Greedy Algorithms Interview Questions.Top 100 DSA Interview Questions Topic-wise.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
